OK apologies for the delay! Here it is:
We developed two phenotypes for adult DM - sensitive cohort ID #302 and specific cohort ID #303.
The sensitive cohort was essentially two separate diagnoses of “dermatomyositis” within one year. We reviewed descendents for each concept ID included in the cohort definition and ensured we were excluding “juvenile” and “childhood” dermatomyositis as our phenotype is focusing on the adult population. We also wanted to exclude “drug-induced dermatomyositis” as this is a different entity than the phenotype of interest.
With the sensitive cohort, the following output was observed across databases (thank you @Gowtham_Rao for the assist):
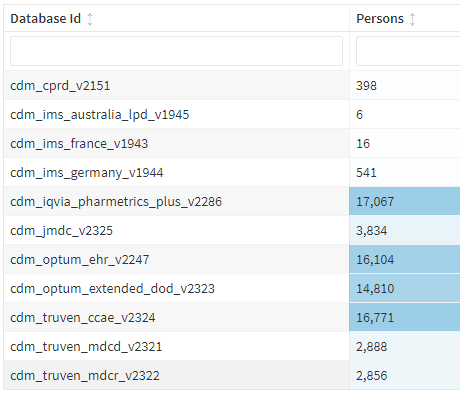
When run against our own Hopkins Myositis Database, this resulted in N=794, which is a very good approximation (we have about 820 adult DM patients enrolled per gold standard chart review).
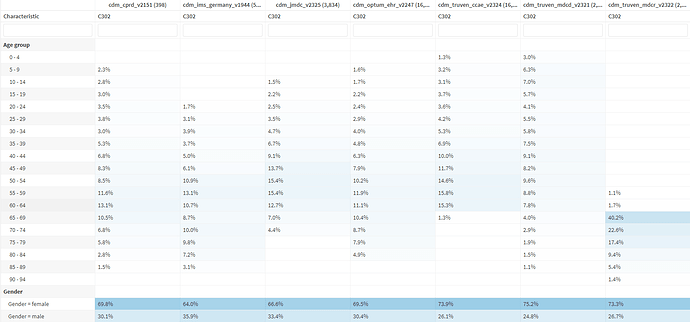
In taking a deeper dive at demographics:
This looks good! We expect this distribution in age and sex given what is known about the epidemiology of adult DM. HOWEVER, clearly there are some patients <18 who are included, which is of course not adult DM. I suspect these are patients with juvenile DM who “aged into” an adult clinic. Alternatively, children with dermatomyositis were simply diagnosed with “dermatomyositis”. We can think about ways of excluding these patients in future iterations of this cohort definition (inclusion >/=18 years old).
One thing that caught my eye is the frequency of the OTHER DM, “diabetes”:
It’s certainly plausible these are real - many older patients have DM, particularly those who have received a lot of prednisone for their adult dermatomyositis. However I do wonder how much cross-reactivity these is between diabetes ‘DM’ and dermatomyositis ‘DM’ from potential coding errors.
With regards to the specific cohort, we attempted to increase specificity by adding requirements for both immunosuppressive medications (used to treat essentially all patients with adult DM) and autoantibodies (adds further confidence of diagnosis, as well as provides a potential pathway to further subgroup dermatomyositis patient populations in future research studies).
However, adding these two inclusion criteria essentially wiped out all cases:
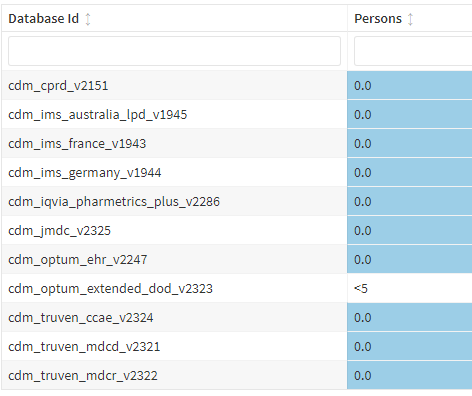
In taking a deeper dive, this appears to largely be related to requiring the antibody test. Based on feedback from our OHDSI calls, there was an idea to separate “test performed” from “test positive”, similar to the approach described in the SLE phenotype this month.
In terms of next steps, we are refining both of our sensitive and specific cohort definitions based on Phenotype Workgroup feedback/Peer Review. The goal will be to finalize these phenotypes within our team, and submit them for further cohort diagnostics and PheValuator with the generous help of @Gowtham_Rao @Joel_Swerdel, @agolozar and others!