Hi all,
I’m trying to use the vocabulary to determine whether ICS drugs should be classified as low/medium/high dose medication for asthma. The amount of active substance is available in the drug_strength table, but what is classified as low/medium/high differs depending on the formulation used. See example table below:
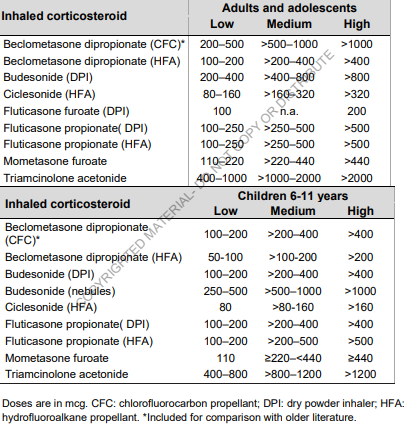
Examples of formulations are:
- CFC = chlorofluorocarbon propellant (not used anymore).
- HFA = hydrofluoroalkane propellant
- DPI = dry powder inhaler
- NEB = nebules
- UFA = ultra fine
- MDI = metered-dose inhaler
I want to link concepts at clinical drug level to these formulations to be able to use these different thresholds. I think dose form can help, but does not cover it entirely. Does anyone know if and how this could be done in OMOP CDM?
@aniekmarkus:
You have two choices:
- You use the DOSE_ERA table, which gives you the dose. But it won’t have the forms
- You build your own cohorts. You can’t do that in ATLAS, because it cannot calculate dose at this time. You’d have to do that in SQL or an analytical packages. When I was in AZ I actually built these. So, can be done.
The formulations you mention: We don’t have that detail. The data just don’t provide it. All you have is inhalants.
Does that help?
@ aniekmarkus
You will have to join the tables where the information is found and in the SQL query you want to create a SQL query with a SELECT statement that uses a CASE statement that will allow you to specify when it is low, medium and high. Let me know if you need help with this and I could help you.
Thanks both of you for your responses. It’s good to know this detail is not in the data and I did not miss it. If I need any further help I will let you know.
